USP
EP
BP
JP
ChP
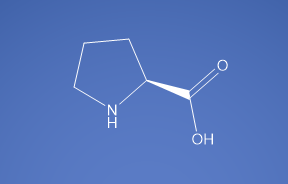
Molecular Formula
C5H9NO2
CAS No
Molecular Weight
Solubility in Water
Boiling Point
Melting Point
Quality Level
Highest – Parenteral Excipient Grade cGMP
Multi-Compendial
Suitable for Injectable Biologic & Vaccine Formulations
L-Proline is a naturally occurring, cyclic imino acid that plays a unique structural role in proteins and is widely used in pharmaceutical manufacturing as both a cell culture component and a formulation excipient.
In upstream biologics production, L-Proline may be incorporated into chemically defined media to support mammalian cell metabolism and recombinant protein synthesis. More prominently, L-Proline is used in downstream formulation of therapeutic proteins—including monoclonal antibodies, recombinant enzymes, and fusion proteins—where it functions as a stabilizing excipient.
Pfanstiehl Pharmaceutical-grade L-Proline is manufactured under cGMP conditions with tight control of purity, endotoxin levels, and trace metal content to ensure consistent performance in regulated drug products.
L-Proline is considered an effective excipient for biologic drug formulations due to its ability to enhance protein stability under thermal, mechanical, and freeze-thaw stress conditions.
As a compatible osmolyte, L-Proline helps maintain protein conformation by preferentially stabilizing the native folded state and reducing aggregation pathways. It can contribute to improved solubility at high protein concentrations and may mitigate viscosity challenges in concentrated monoclonal antibody formulations. In addition, L-Proline can support isotonicity and buffer systems without negatively impacting protein structure or biological activity.
The use of highly purified L-Proline is essential in biologic formulations, as trace impurities, endotoxins, or metal contaminants can promote oxidative degradation, aggregation, or variability in product quality attributes.
Pfanstiehl Pharmaceutical-grade L-Proline supports robust formulation development, improved shelf stability, batch-to-batch consistency, and alignment with global regulatory expectations for biologics manufacturing.
was developed specifically for biopharmaceutical manufacturing and is produced under full ICH-Q7 cGMP conditions in the United States to the highest quality and purity standards.
Pfanstiehl Brand Multi-Compendial L-Proline is compliant with USP EP BP JP and ChP pharmacopoeia.
Because of these stringent manufacturing specifications and Pfanstiehl quality systems, you can be assured that Pfanstiehl’s Tris Base will be of the highest quality & consistency from batch to batch and the safest available for use in your manufacturing or research application.
L-Proline
Multi-compendial
GMP
Injectable Excipient Grade
USP
EP
BP
JP
ChP
Parenteral Formulation
L-Proline
Multi-compendial
GMP
Injectable Excipient Grade
USP
EP
BP
JP
ChP
Parenteral Formulation