Highest Purity
Lowest Endotoxin
Lowest Trace Metals
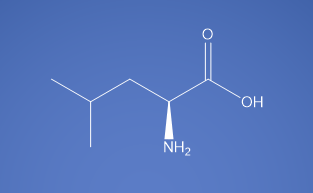
Molecular Formula
C6H13NO2
CAS No
Molecular Weight
Solubility in Water
Boiling Point
Melting Point
Quality Level
Highest – GMP Injectable Excipient Grade
Multi-Compendial
L-Leucine is a branched-chain essential amino acid widely used in pharmaceutical manufacturing as both a process aid and functional excipient. In biologics production, L-Leucine may be incorporated into chemically defined cell culture media to support protein synthesis and cellular metabolism in mammalian expression systems. More prominently, L-Leucine is utilized in drug formulation—particularly in spray-dried and particle-engineered dosage forms—where its physicochemical properties contribute to improved powder performance.
Pfanstiehl Pharmaceutical-grade L-Leucine is manufactured under controlled conditions to ensure high purity, low endotoxin levels, and minimal trace metal content, supporting consistent performance in regulated drug products.
L-Leucine is considered an especially valuable excipient for inhalation drug applications, including dry powder inhalers (DPIs), due to its ability to enhance aerosolization and dispersibility. During spray drying, L-Leucine tends to migrate to the particle surface, forming a crystalline shell that reduces interparticle cohesion and moisture uptake. This surface enrichment improves powder flow, minimizes aggregation, and increases fine particle fraction—critical parameters for efficient deep lung delivery.
Additionally, L-Leucine can improve formulation stability by reducing hygroscopicity and enhancing physical robustness during storage and handling. The use of highly purified L-Leucine is essential in inhalation products, where impurities or particulate contaminants could compromise aerodynamic performance, device compatibility, product stability, or patient safety.
Pharmaceutical-grade L-Leucine enables reliable particle engineering, consistent dose delivery, and regulatory compliance in advanced pulmonary drug delivery systems.
was developed specifically for biopharmaceutical manufacturing and is produced under full ICH-Q7 cGMP conditions in the United States to the highest quality and purity standards.
Pfanstiehl Brand Multi-Compendial L-Leucine is compliant with USP EP BP , JP, and ChP pharmocopoeia.
Because of these stringent manufacturing specifications and Pfanstiehl quality systems, you can be assured that Pfanstiehl’s L-Leucine will be of the highest quality & consistency from batch to batch and the safest available for use in your manufacturing or research application.
L-Leucine
Multi-Compendial
GMP
Highest Purity
Lowest Endotoxin
Lowest Trace Metals
USP
EP
BP
JP
ChP
Parenteral Formulation
Spray Dry Formulation
Lyophilization Formulation
L-Leucine
Multi-Compendial
GMP
Highest Purity
Lowest Endotoxin
Lowest Trace Metals
USP
EP
BP
JP
ChP
Parenteral Formulation
Spray Dry Formulation
Lyophilization Formulation