- Highest Purity
- Lowest Endotoxin
- Lowest Metals
- Lowest Bioburden
- Lowest Nitrosamines
Menu
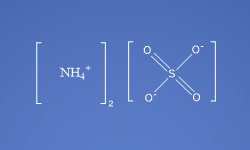
Molecular Formula
(NH4)2SO4
CAS No
Molecular Weight
Solubility in Water
Boiling Point
Melting Point
Quality Level
Highest – GMP Injectable Excipient Grade
Manufactured under ICH-Q7 GMP regulation
Ammonium sulfate ((NH₄)₂SO₄) is an inorganic salt widely used in biopharmaceutical and pharmaceutical manufacturing for protein purification, process development, and certain formulation and analytical applications.
In biologics production, ammonium sulfate is most commonly utilized in downstream processing as a precipitation agent to selectively isolate and concentrate proteins through controlled “salting-out.” By increasing ionic strength, ammonium sulfate reduces protein solubility in a predictable and tunable manner, enabling fractionation based on differences in protein surface properties. This approach is frequently employed in early purification steps to remove host cell proteins, concentrate target biomolecules, or stabilize intermediates prior to chromatographic polishing. In addition, ammonium sulfate is used in buffer systems and laboratory-scale process development where controlled ionic environments are required.
For pharmaceutical applications, the purity of ammonium sulfate is critical to ensuring process consistency, product quality, and regulatory compliance. Trace metals, residual contaminants, insoluble particulates, or microbial endotoxins can negatively impact protein stability, promote aggregation, interfere with chromatography performance, or introduce variability into sensitive biologic manufacturing processes. Impurities may also contribute to filter fouling, reduced resin lifetime, or unintended modifications of protein structure.
Pfanstiehl high-purity, low-endotoxin, low-metals ammonium sulfate supports robust downstream processing, minimizes batch-to-batch variability, and aligns with cGMP manufacturing requirements and global regulatory expectations. By using pharmaceutical-grade ammonium sulfate, manufacturers enhance process control, protect product integrity, and reduce risk across the biologics production lifecycle.
was developed specifically for biopharmaceutical manufacturing and is produced under full ICH-Q7 cGMP conditions in the United States to the highest quality and purity standards.
Because of these stringent manufacturing specifications and Pfanstiehl quality systems, you can be assured that Pfanstiehl’s Ammonium Sulfate will be of the highest quality & consistency from batch to batch and the safest available for use in your manufacturing or research application.
Ammonium Sulfate
Highest Purity
Lowest Endotoxin
Lowest Metals
GMP
NF
ChP
Parenterals
Liquid Formulation
Biopharma process
Ammonium Sulfate
Highest Purity
Lowest Endotoxin
Lowest Metals
GMP
NF
ChP
Parenterals
Liquid Formulation
Biopharma process